 implement a UDI system in accordance with Article 24, 26, and 28.CE Mark the product in accordance with Article 18.create and maintain a Declaration of Conformity in accordance with Article 17.create and maintain a technical file in accordance with Annex II & III.This requirement is defined in Article 52 and Annex XIII conduct a performance evaluation–including a post-market performance follow-up (PMPF).a risk management process in accordance with Annex I – deviations from ISO 14971:2019 will be necessary).The Technical File requirements are found in Annex II and III while most of the quality system requirements are found in the Articles. The notified body must also confirm that their quality system has been updated to include the IVDR requirements. Under the IVDR, a notified body must now review and approve the technical file. Most of the companies that are preparing for an IVDR audit by their notified body already have ISO 13485:2016 certification and they are using the self-declaration pathway for CE Marking under the IVDD. In the fourth column the auditor will enter the objective evidence for conformity collected during the audit. Some of the information in the second and third columns can often be entered prior to starting the audit by reviewing audit preparation documents (e.g. 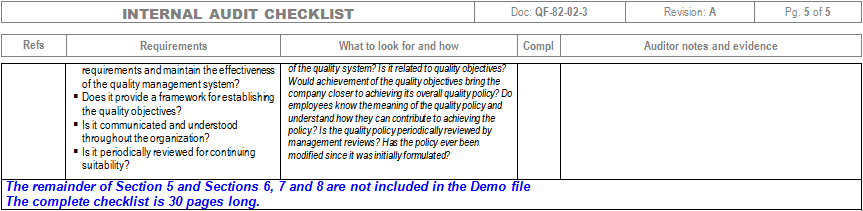
In the third column, the auditor indicates what they were looking for in the records, procedures, or during the interview. The next column is where the auditor documents records sampled, procedures reviewed, and personnel interviewed. The left-hand column lists each requirement. Typically the checklist is in four columns using a tabular form. If the auditor is unable to find supporting evidence of compliance, the auditor may continue to search for data or identify the requirement as a nonconformity. This objective evidence verifies compliance with regulatory requirements or internal procedural requirements. If you would like to download our IVDR checklist for FREE, please fill in the form below.Īn audit checklist is used by quality system auditors to collect objective evidence during an audit. Usually, we conduct internal audits using the process approach to auditing, but this is one of the scenarios where the element approach and an audit checklist are invaluable. Therefore, IVD manufacturers are asking Medical Device Academy to conduct remote internal audits of their quality system to identify any gaps. 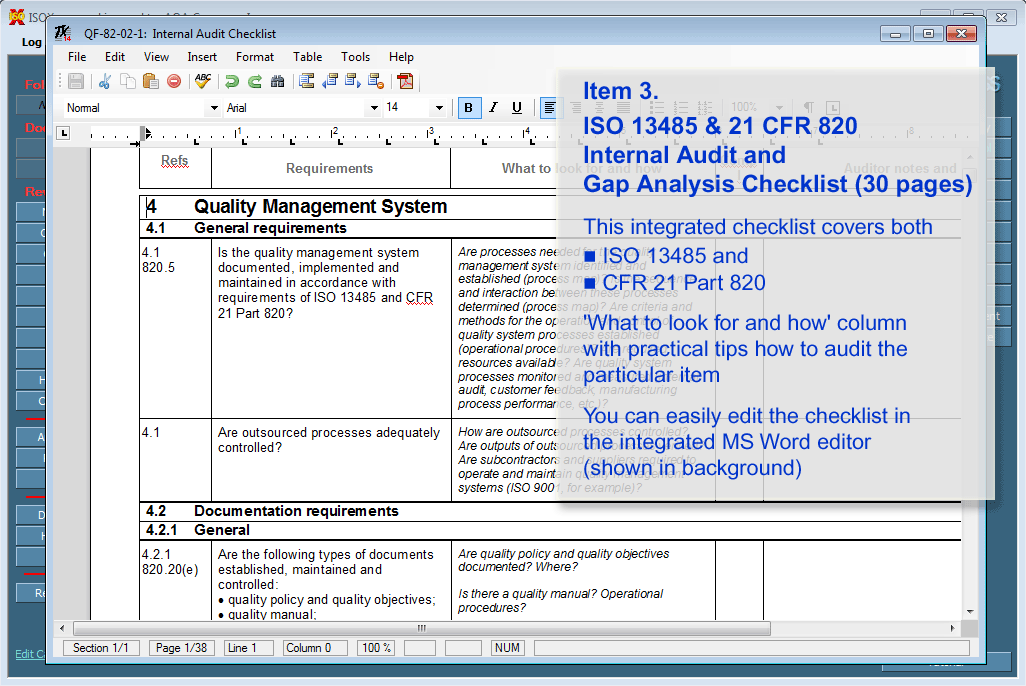
However, there is much more that needs to be updated than just the technical documentation. Revision of technical files and the associated procedures for creating your technical files is a big part of these updates. 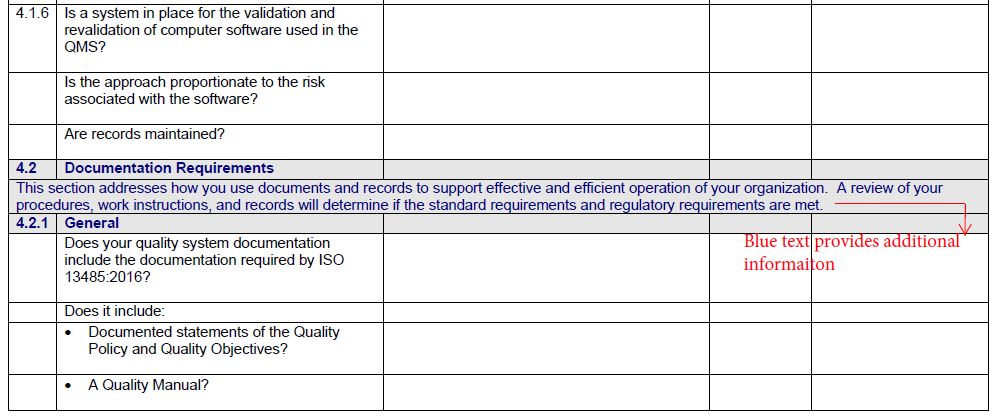
Directive 98/79/EC) or IVDD to the new EU In Vitro Diagnostic Regulation (i.e. Hundreds (if not thousands) of IVD manufacturers are currently updating their ISO 13485:2016 certified quality system from compliance with the In Vitro Diagnostic Directive (i.e. This article provides an IVDR checklist for updating your ISO 13485 quality system to comply with EU Regulation 2017/746. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
